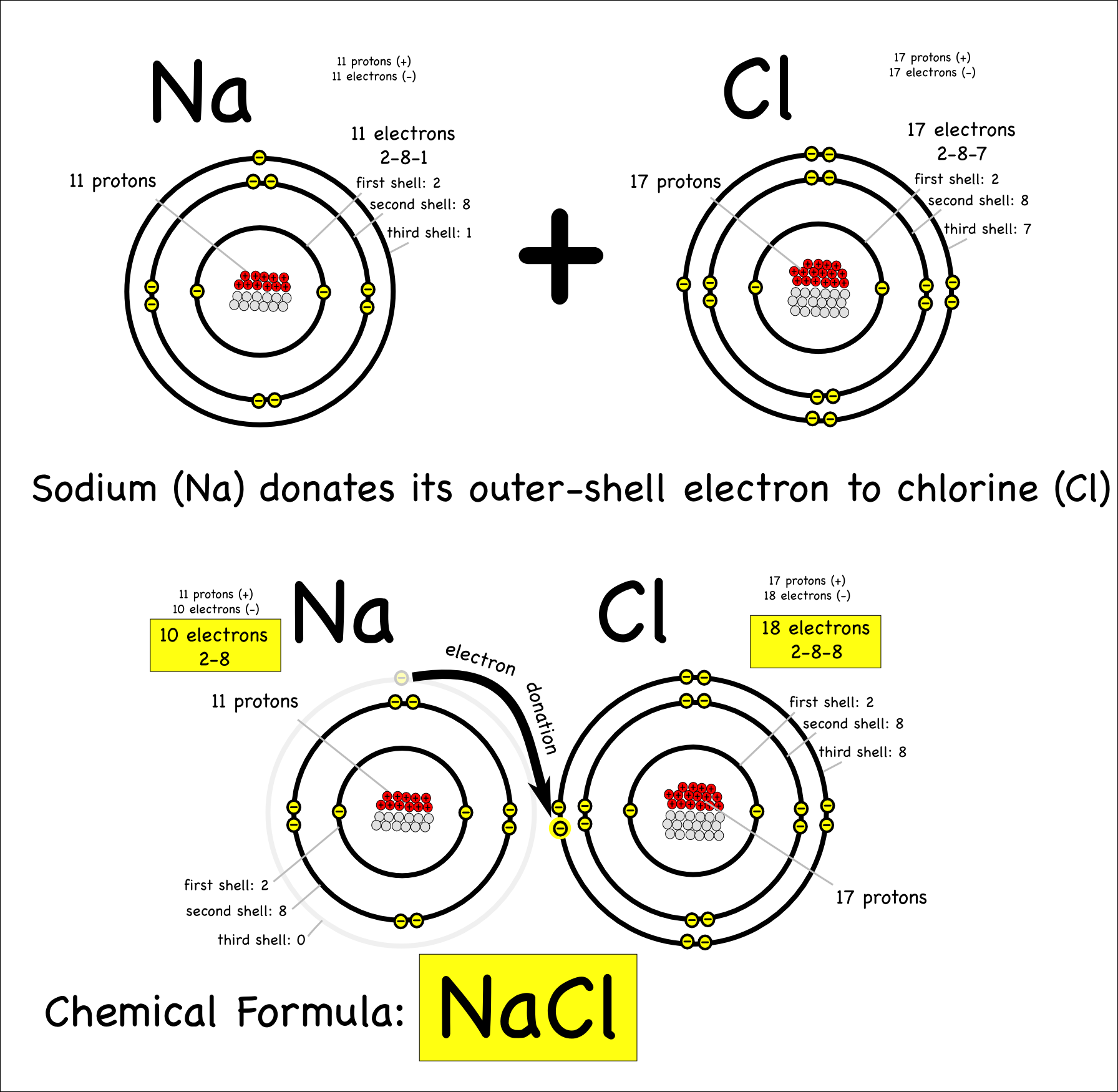
According to Coulombs law, the attraction of an electron to a nucleus depends only on three factors: the charge of the nucleus (+Z), the charge of the electron (-1), and the distance between the two ((r)). An element symbol without a charge written next to it is assumed to be the uncharged atom. The ideal gas law is easy to remember and apply in solving problems, as long as you get the proper values a. If the charge is a single positive or negative one, the number 1 is not written if the magnitude of the charge is greater than 1, then the number is written before the + or − sign. The magnitude of the charge is listed as a right superscript next to the symbol of the element. Note the convention for indicating an ion. Note: The blank area shown in the above periodic table are mostly the transition and post-transition elements. In these compounds, halogens are present as halide anions with charge of -1 (e.g., Cl-, Br-, etc.). All halogens form Group 1 salts with similar properties. As a general rule, fluorine is the most reactive halogen and astatine is the least reactive. Hence the ionic charge of Chlorine (Cl) is 1-. However, halogens readily combine with most elements and are never seen uncombined in nature. Unfortunately, there is little understanding which two charges a metal atom may take, so it is best to just memorize the possible charges a particular element can have. Elements of group 17 have 1-ionic charge, Elements of group 18 have 0 ionic charge, Now here our element is Chlorine (Cl) which lies in group 17 of the periodic table. Cobalt (Co) is another element that can form more than one possible charged ion (2+ and 3+), while lead (Pb) can form 2+ or 4+ cations. 1: A trick for writing electron configurations based on the organization of the periodic table. For example, iron (Fe) atoms can form 2+ cations or 3+ cations. A few elements, all metals, can form more than one possible charge. Examination of the table in the link given shows that there are some exceptions to the previous point. A more complete table of ions and their charges can be found at Monotomic Ions of Various Charges. The symbol for the ion is N 3−, and it is called a nitride ion.\) is helpful in determining the charge on a large number of our cations and anions it's hardly complete. Thus, a nitrogen atom will form an anion with three more electrons than protons and a charge of 3−. A nitrogen atom must gain three electrons to have the same number of electrons as an atom of the following noble gas, neon. Nitrogen’s position in the periodic table ( group 15) reveals that it is a nonmetal. It has a + 1 charge, because there are 11 protons in the nucleus, but only 10 electrons around the nucleus of the ion. The usual charge of an element is common to its group. The symbol for the ion is Mg 2+, and it is called a magnesium ion. There are four ways to find the charge of an element: Use the periodic table. The formula for sodium chloride, therefore, is NaCl since Na loses one electron and has a +1 charge and Cl gains one. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. A magnesium atom must lose two electrons to have the same number electrons as an atom of the previous noble gas, neon. Cl, N, P), and 0 for each carbon atom bonded. Magnesium’s position in the periodic table ( group 2) tells us that it is a metal. The oxidation state of an atom is the charge of this atom after ionic approximation of its heteronuclear bonds. Write the symbol for each ion and name them. Predict which forms an anion, which forms a cation, and the charges of each ion. Magnesium and nitrogen react to form an ionic compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
